Call :
PENTA Call 1
Project status :
Completed
Project website :
http://serene.minalogic.net/
Key application areas
 Health & Well-Being
Health & Well-Being  Energy
Energy  Digital Industry
Digital Industry  Digital Life
Digital Life Essential capabilities
 Systems and Components Architecture, Design & Integration
Systems and Components Architecture, Design & Integration
Connectivity & Interoperability
 Safety, Security & Reliability
Safety, Security & Reliability  Computing & Storage
Computing & Storage  ECS Process Technology, Equipment, Materials & Manufacturing
ECS Process Technology, Equipment, Materials & Manufacturing
PARTNER
Autonomous University of Barcelona / CEA / Centre Hospitalo-Universitaire Grenoble Alpes / FLAVIA IT-Management GmbH / FRESENIUS-VIAL SAS / Fraunhofer EMFT / IDEMIA France SAS / Klinikum der Universitaet Muenchen / LCIS / Institut Polytechnique de Grenoble / MAATEL SAS / Medtronic - Sofradim Production / Morpho / Orange Labs / SGS-TÜV Saar GmbH / STMicroelectronics / Sensing Tex / X-FAB Dresden GmbH & Co. KG / spring techno GmbH Co KG
Autonomous University of Barcelona / CEA / Centre Hospitalo-Universitaire Grenoble Alpes / FLAVIA IT-Management GmbH / FRESENIUS-VIAL SAS / Fraunhofer EMFT / IDEMIA France SAS / Klinikum der Universitaet Muenchen / LCIS / Institut Polytechnique de Grenoble / MAATEL SAS / Medtronic - Sofradim Production / Morpho / Orange Labs / SGS-TÜV Saar GmbH / STMicroelectronics / Sensing Tex / X-FAB Dresden GmbH & Co. KG / spring techno GmbH Co KG
Countries involved
 Spain
Spain  France
France  Germany
Germany Project leader(s)
Armand CASTILLEJOKey project dates
01 June 2017 - 30 November 2020SERENE-IoT addressed the needs of patients being followed remotely by healthcare professionals by developing, in Europe, advanced smart e-health IoT medical devices & an end-to-end architecture.
The SERENE-IoT project contributed to the development of high-quality connected healthcare services and diagnostic tools based on advanced smart healthcare Internet of Things (IoT) devices, fully manufactured in Europe. It thereby enabled a high level of ‘quality of service’ for patients being followed remotely by professional caregivers, and at a lower cost than traditional care provided today.
Background, objectives of the project and challenges
Regarding the introduction of IoT technologies, the major project objectives were:
1) development of three clinical prototypes of new medical devices supporting security, safety and privacy, and their complete validation within a secured end-to-end IoT system platform
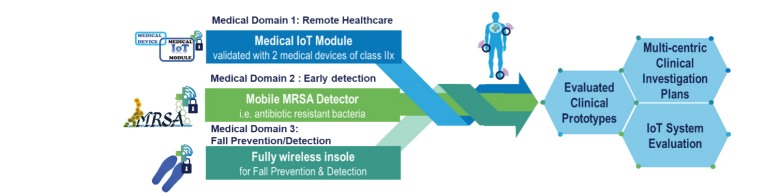
2) provision of clinical investigation plans associated with each device
3) implementation of a secured IoT system platform to validate the three prototypes in real-world scenarios, including the proposed healthcare data structure
4) demonstration and evaluation of the benefits obtained from this new secure, remote healthcare control and monitoring system. It should be noted that a reduction in healthcare expenses was expected.
These objectives will lead to new market opportunities for the European healthcare industry by establishing a basis for the creation of new business models.
SERENE-IoT addressed three main medical challenges in the following domains:
- Remote healthcare – by moving care services from hospital to home and investigating two medical use-cases: (1) the first wearable low-power ‘Medical IoT Module’, bringing connectivity to connected infusion / nutrition pump systems in Medical Device class IIx, and (2) a post-surgery infection detection system
- Early detection – investigating a methicillin-resistant bacteria (MRSA) mobile diagnostic device
- Fall prevention – by investigating wireless insoles for gait analysis and fall prediction
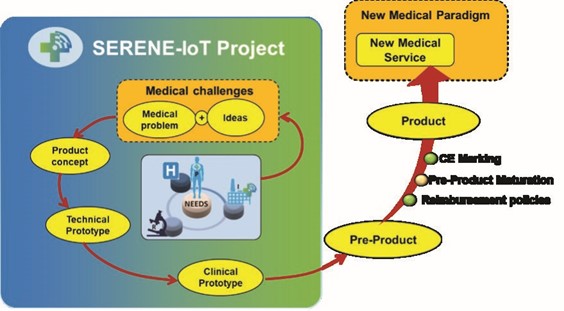
In line with the medical innovation cycle up to the clinical prototype stage, three devices, one per domain (as described below), were developed and subsequently validated by healthcare professionals over the course of the project. The certification and industrialisation phases will follow the end of SERENE-IoT project under the responsibility of the relevant MedTech OEM. The main technology aspects addressed by the project were low-power, multi-protocol connectivity; end-to-end system security; interoperability; and more adequate computing power.
Technological achievements
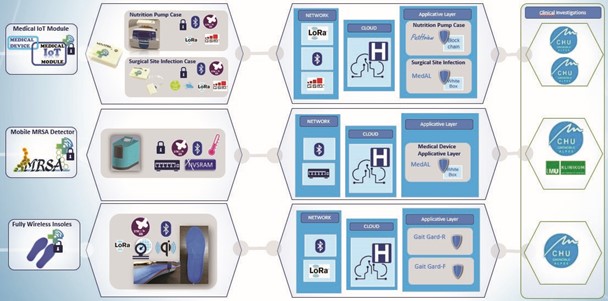
SERENE-IoT delivered the three following technical prototypes:
- A Medical IoT Module, investigated in two different medical devices (class IIx)
- A connected methicillin-resistant staphylococcus aureus (MRSA) detector
- A pair of wireless insoles for fall prevention
The three technical prototypes were integrated into an installation, along with the relevant dedicated platforms, at the Grenoble University Hospital (CHUGA). The complete solution, dedicated to real world use of the three prototypes, was installed in the hospital infrastructure to carry out an end-to-end demonstration in clinical settings with dedicated application layers. The Medical IoT Module was integrated with two different application layers: ‘PatHView’ dedicated to the nutrition pump case, and ‘MedAL’ for the surgical site infection (SSI) detection case. MedAL was also used with the connected MRSA detector. For the wireless insoles, the ‘Gait-F’ application layer for fall prevention was also duplicated into a ‘Gait-R’ version which was implemented in a rehabilitation use-case proposed thanks to the project collaboration.
All three technical prototypes received feedback from clinicians to enable continuation of the medical innovation cycle.
The Medical IoT Module received feedback from the artificial nutrition unit and the digestive surgery unit (of CHUGA) regarding both use cases (the nutrition pump and the SSI case). Clinical investigations on real patients were still running at the end of the SERENE-IoT project in the nutrition pump case (Medical IoT Module), highlighting the strong level of clinician interest in this prototype. The connected MRSA detector received feedback from the Hospital Hygiene Dept (CHUGA) and from KUM (University Hospital of Munich), while the wireless insoles received feedback from the Geriatrics and Ortho-geriatrics departments in CHUGA. For all the prototypes, projects are being finalised to continue the next steps of the medical innovation cycle.
All the pre-clinical and clinical evaluations have provided relevant functional, usability and clinical feedback to manufacturers by focusing on critical points. This will allow improvement of these devices and development of final products optimised for clinical usage. Usually, six years are needed to go from initial idea to first prototype. In the SERENE-IoT project, this time has been cut in half.
Thanks to the project, academic partners have also offered access to a platform that allows for the exploration of security issues. The platform illustrates security threats on medical devices which do not have proper countermeasures and validates software and hardware countermeasures for specific use cases (such as SecPump developments).
Market Potential
The MedTech partners in the SERENE-IoT MedTech project are active in the global IoT in healthcare market, a market expected to grow from USD 72.5 billion in 2020 to USD 188.2 billion by 2025 (CAGR:21.0%). The Medical IoT Module from Maatel addresses this full spectrum. During the project, it was investigated in two different medical devices (a nutrition pump from Fresenius consolidating their position in this market, and a post-operative surgical site infection (SSI) device from Medtronic creating a new market opportunity). The second prototype, the MRSA detector will create new market opportunities in the diagnosis market (CAGR 10.5%) while enabling the creation of a start-up. Thirdly, the wireless insoles will create new market opportunities for SensingTex in various market segments (elderly, rehabilitation …) with a CAGR from 4 to 20%.
High-tech project partners will obviously benefit from these market opportunities for their hardware or software building blocks, but also from other application domains where healthcare is just one of the possible verticals (even if it represents a less and less negligible part over the years). The technology markets concerned are Bluetooth Low Energy, embedded security, non-volatile (NV) memory, software security and IT.
Societal & Economic Impact
From the project point of view, SERENE-IoT has impacted most of the partners, with around 10 people hired in their teams.
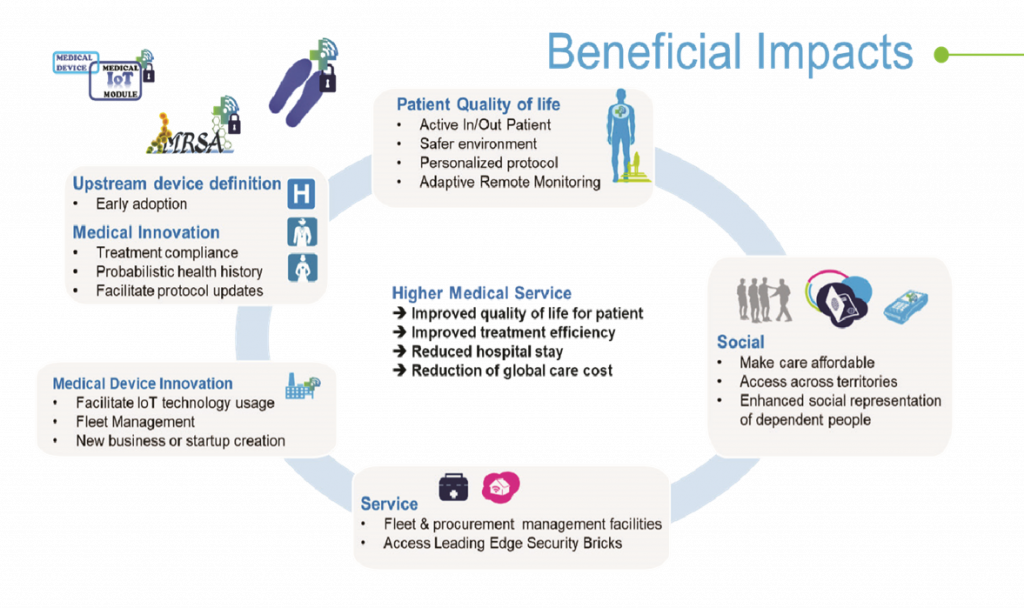
In addition, all the stakeholders around the patient and patients themselves are expected to derive benefits from SERENE-IoT developments.
Patients are expected to receive remote follow-up that results in reduced hospital visits or stays. By being at home with their family, work, etc …, the patient can benefit from an improved quality of life along with a high level of quality of care. They can be treated effectively in their normal home / work environment.
Thanks to the organisation of care enabled by the SERENE-IoT technology, Medtech partners estimate cost savings of between 25 and 50% depending on the medical use case.
One of the most important impacts of SERENE-IoT is the impressive acceleration enabled by the project. The medical innovation cycle is quite long – it usually requires 10 years to bring a new service into medical practice. SERENE-IoT provided feedback on clinical prototypes after three years instead of the six years typically needed.
The recent outbreak of COVID-19 has led IoT healthcare solution providers to quickly deliver solutions to tackle the rising demand for high-quality remote services. Applications, such as telemedicine, remote patient monitoring and interactive medicine, along with in-patient monitoring, are expected to gain traction during this time.
The new Covid-19 reality is accelerating drivers to digital transformations in healthcare:
- Telemedicine demonstrates industry-altering benefits
- Patient care involvement
- Acceptance of technologies & demand from patients & doctors
- Shortages of healthcare professionals
- Budget pressures
- Dynamic medical regulations updates
- Governmental funding initiatives around the world
| Patents/Standardisation/ Publications SERENE-IoT achieved 7 patents and engaged in 60 dissemination opportunities during project period: Patents: Idemia(2), CEA(2), LCIS(2), ST(1)IEEE publications: 11 Other publications: 15 Conference/Talks: 29 Public Webinars: 5 Future Developments Several further developments are ongoing. First, benefitting from the SERENE-IoT acceleration, its Medtech partners will pursue the remaining steps of the medical innovation cycle to bring their products and services into medical practice. In the short term, several projects will be submitted to continue clinical investigations (MRSA detector, wireless insoles). STMicroelectronics is continuing to support the medical partners in the use of its technologies, helping to structure the European healthcare value chain. With its R&D and medical application ecosystem, STMicroelectronics is looking to submit a new acceleration initiative currently named SERENE-AI. |






