Call :
PENTA Call 2
Project status :
Completed
Project website :
http://ulimpia-project.eu/
Key application areas
 Health & Well-Being
Health & Well-Being  Digital Industry
Digital Industry  Digital Life
Digital Life Essential capabilities
 Systems and Components Architecture, Design & Integration
Systems and Components Architecture, Design & Integration
Connectivity & Interoperability
 Safety, Security & Reliability
Safety, Security & Reliability  Computing & Storage
Computing & Storage  ECS Process Technology, Equipment, Materials & Manufacturing
ECS Process Technology, Equipment, Materials & Manufacturing
PARTNER
Delft University of Technology / Fraunhofer EMFT / Fundació Eurecat - Technology Centre of Catalonia / GED Gesellschaft für Elektronik und Design mbH / Henkel AG and Co KGaA / IMEC / Institutes of Textile and Fiber Research Denkendorf (DITF) / KOB GmbH / LINXENS France / MEPY BENELUX / NXP Semiconductors Germany GmbH / Novioscan BV / Philips Electronics Nederland BV / Picosun Oy / TNO / VTT Technical Research Centre of Finland Ltd / warmX GmbH
Delft University of Technology / Fraunhofer EMFT / Fundació Eurecat - Technology Centre of Catalonia / GED Gesellschaft für Elektronik und Design mbH / Henkel AG and Co KGaA / IMEC / Institutes of Textile and Fiber Research Denkendorf (DITF) / KOB GmbH / LINXENS France / MEPY BENELUX / NXP Semiconductors Germany GmbH / Novioscan BV / Philips Electronics Nederland BV / Picosun Oy / TNO / VTT Technical Research Centre of Finland Ltd / warmX GmbH
Countries involved
 Netherlands
Netherlands  Germany
Germany  Belgium
Belgium  France
France  Finland
Finland  Spain
Spain Project leader(s)
Ad de BeerKey project dates
01 April 2018 to 31 March 2021Ulimpia has created the world’s first smart, wearable ultrasound patch for diagnosis and health monitoring at home, using an open platform approach.
ULIMPIA’s MEMS (Microelectromechanical systems)-based technology can continuously monitor and diagnose processes under the skin or even deep within the body, such as blood pressure or bladder function. So, more patients can stay at home, reducing hospital costs. In addition, ULIMPIA’S open platform allows other developers to create new applications for the growing consumer healthcare market.

From artist impression to realisation. Copyright Philips
Background, objectives of the project and challenges
Faced with aging populations and rising chronic disease, healthcare systems worldwide are looking for solutions to improve care and limit costs. ULIMPIA aimed to address these needs by developing point-of-care monitoring and diagnostic solutions that enable affordable continuous on-body monitoring and empower patients to better manage their health at home. The project partners ranged from leading companies in the fields of semiconductor devices (MEMS, ASIC design), materials and packaging, medical devices and wearables manufacturing to world-class medical institutions. The technological challenge was to create the world’s first universal programmable ultrasound engine building block. This engine would be the basis for an open platform (in the form of a body patch) that would be accessible to multiple users, and which could lead to an industry standard. Achieving these ambitions called for innovation in areas from MEMS and ASICs to yarns and materials tailored to specific end-user requirements and specific algorithms. The electronics on the platform had to carry out sensing reliably, and the patch had to be adhesive, stretchable and long-life but non-irritating to the skin.
The project envisaged numerous potential applications: blood pressure measurement, bladder monitoring, inspection of blood vessels in diabetes patients, early breast cancer detection, needle guidance and wound monitoring. Its work resulted in a demonstration body patch platform that could be wirelessly connected to a peripheral device such as a mobile phone. The mobile device then analysed and displayed the data to give real-time feedback to the user.
ULIMPIA achieved this successful result despite the constraints of the COVID-19 pandemic. A combination of strong cooperation and detailed planning helped the partners involved collaborate effectively even under such difficult conditions.
Technological achievements
Ultrasound-based continuous on-body monitoring is a major technological breakthrough from the Ulimpia project, delivering several key innovations:
- Programmable ultrasound module
- Universal controller, power management and wireless connectivity module (Android app)
- Conformable patch technology connecting one or more ultrasound modules to the universal controller module
- Flexible/knitted interconnect.
Ulimpia developed and prototyped a 2D version of the ULIMPIA ultrasound module including a front-end ASIC and supporting FPGA. Bluetooth Low Energy (BTLE) provided connectivity to back-end applications. The battery-powered patch platform was implemented in various demonstrators with sensors and interconnectors, showing the potential of the modular approach and innovations such as gel-free contact with the skin. Specially created 20-30 µm polyamide multi-filament yarns with silver coating were integrated into the demonstrators together with a purpose-designed interposer housing.
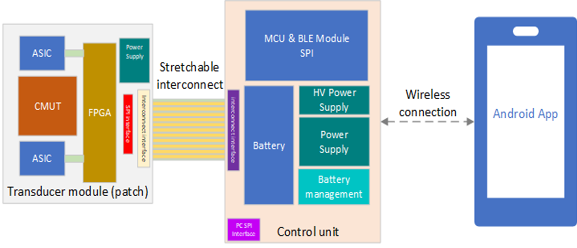
Block diagram of the Ulimpia patch showing the individual parts: transducer module, control unit, stretchable interconnect and the android app.
One demonstrator focused on novel wound condition monitoring capability for long-term chronic wounds, integrating pH and wound temperature sensors, basic electronic interconnects and BTLE connectivity, a specially developed integrated moisture sensor and an engineering-level Android app.

Pictures, showing the wound monitoring patch, are taken form the public Deliverable, D6.4.
The partners assessed a smart comfort patch made with knitted yarns placed on a ‘skin phantom’ exploring monitoring vasomotor symptoms and development of a calorimeter to measure human body heat generation. Uncorrupted data readings were obtained even when sensors are close to the warmth of the body, a vital precursor for moving to clinical trials.
The project successfully measured blood pressure, comparing the performance of two emerging technologies: CMUT and PMUT. It conducted a ‘phantom’ measurement of adult bladder fullness using CMUT prototypes to a good level of accuracy.
An unforeseen application arose – COVID testing. This use case provided an early indicator of the value of the project’s open approach.

Ulimpia patch used as bladder monitor on a Phantom from Novioscan.
Picture is taken from public Ulimpia movie.
Market Potential
ULIMPIA’s results address growing global trends. Personal/portable ultrasound technology is emerging, with early market entrants demonstrating systems using Edge AI and cloud-based services. There are moves towards the decentralization of healthcare, personalized therapies and individualized, self-administered diagnostics. COVID-19 accelerated these trends, increasing demand for smart, low-cost healthcare devices for easy use at home. The market for medical wearables is predicted to grow by a CAGR of 40%. By bringing ultrasound to this market, ULIMPIA is expected to drive high production volumes and fuel further innovation in MEMS technologies to reinforce Europe’s strength in professional ultrasound diagnostics. As MEMS-based ultrasound matures it will compete with current more expensive technologies, creating market opportunities for low power, reduced data rate wearable ultrasound devices driven by AI algorithms that interpret data without the need for a medical specialist.
Societal & Economic Impact
The emergence of personal / portable ultrasound has been enabled by innovations in areas such as MEMS, Edge AI and cloud technologies. Meanwhile, the applications focus on pressing needs within healthcare systems worldwide. With an ageing population and rising rates of chronic diseases such as cardiac vascular diseases (CVD), obesity, diabetes and chronic obstructive pulmonary disease (COPD), Europe alone is facing significant increases in healthcare costs – from USD 383 billion in 2016 to an expected USD 425 billion in 2025. The COVID-19 pandemic has only increased these pressures.
It is these demands that are driving the shift towards more home-based care; lower-cost diagnostics that need less professional involvement; personalized therapies; and payments based on cure and prevention, rather than simply treatment.
ULIMPIA has contributed to potential solutions by opening the way to relatively low-cost, mass-producible, portable and wearable ultrasound devices that support the needs of both patients and healthcare systems.
Currently, opportunities are being explored for further development of the Ulimpia platform. First discussions with potential partners have at least resulted in enthusiastic reactions and very positive feedback to the Ulimpia achievements. These discussions will be followed-up soon.
Patents/Standardisation/ Publications
The ULIMPIA project has been presented at more than 39 conferences and symposia and lead to 7 peer reviewed academic papers, 4 articles as well as other communications such as videos for the general public. In total more than 50 publications were realized during the Ulimpia project, while more publications are expected after the end of the project. Next to this several announcements on the Ulimpia results have been posted on social media, in particular on LinkedIn.
Future Developments
The work will continue on integration, miniaturization and power reduction to achieve a small, unobtrusive patch on a coin-sized battery. Efforts will focus on improving signal quality for deep-in-body monitoring and on cloud and app connectivity for data collection and continuous patient monitoring. New projects are being explored such as continuous thorax monitoring for heart failure and remote, non-invasive monitoring of mothers and babies before and after birth in at-risk pregnancies. The latter would be based on a multi-sensor patch featuring ULIMPIA CMUT and patch technologies.

Newlife initiative- New remote non-invasive solutions for ensuring the health of mothers and babies before and after birth







